Propose an explanation for why this occurred.ġ. What changes occurred in the water level when you put the Erlenmeyer over the flask. What did you observe that helped you decide this?Ĥ. Do you think that gas would be oxygen, hydrogen or carbon dioxide? Some fire extinguishers have a compressed gas in them. What gas in our atmosphere promotes combustion?ģ. What are two products from the combustion of a candle?Ģ. List at least two observations/activities that helped you determine this and explain how it helped you decide this.Ģ. Do your results indicate that the candle burns as a solid, liquid or a vapor? What does this tell you about the gas that was formed?ġ. Stopper the flask and swirl the solution for about 1 minute. Add 10 ml of phenol red solution to the flask. After 1-2 minutes, lift the flask out of water and quickly place the rubber stopper in the mouth of the flask. Hold the flask in place for approximately one minute. Quickly lower the mouth of the Erlenmeyer flask over the candle so that the mouth of the candle is below the surface of the water. Pour water in to the pan that the candle is in to a depth of 1cm. What do you think the compound is? Where do you think that the compound came from?ħ. If you see soot, you are probably holding the candle too close to the flame. That is not the compound that we are looking for. 
Note: you may see the formation of black soot on the bottom of the beaker. Look for the formation of a new compound on the bottom of the beaker. Hold the beaker about 4-5 cm above the flame. Place approximately 150 ml of ice water in a 250 ml beaker. What do you thick is happening? What does this say about the part of the candle that is burning?Ħ. If the flame appears to go out, quickly move the wire gauze to the side. Using a tongs, hold the wire gauze over the flame, perpendicular to the flame. Relight the standing candle and blow out the second candle. What does this tell you about the part of the candle that is burning?ĥ. Do you have to touch the wick in order to get the candle to relight? Write your observations below. Gently blow out the first candle flame and then move the other flame in to the smoke from the first flame. Light a second candle and hold the flame 2-4 cm from the flame of the first candle. _wax burning and forming carbon dioxideĤ. Classify each of the following as a chemical (C) or physical (P) change.ī. 
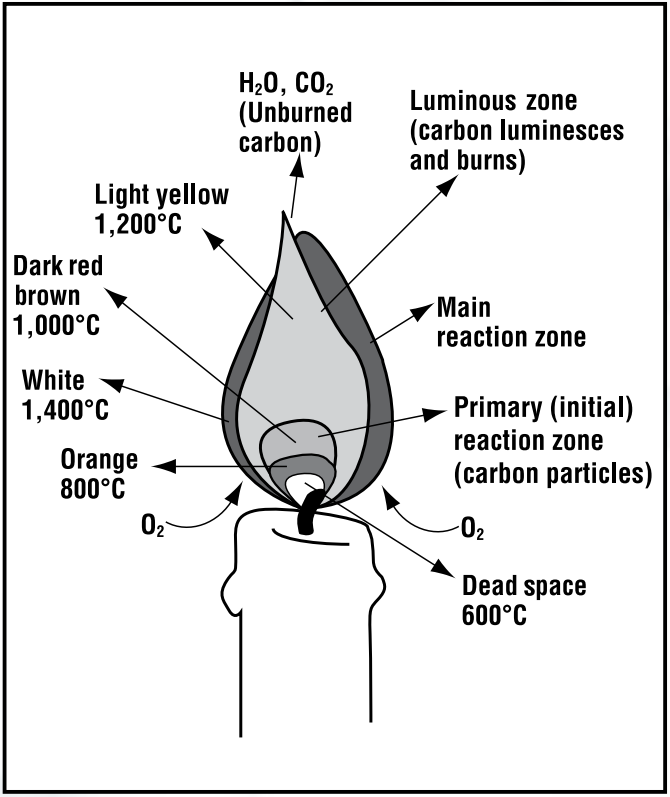
When a physical change takes place, the substance changes shape or phase, but the substance remains the same. When a chemical change takes place, a different substance is formed. Where does the burning take place? What is burning?ģ. Look at the colors and the shape of the flame. List at least 8 observations of the flame. Press the candle into the melted wax and hold it upright until the wax solidifiesĢ. Light a candle and let 3-4 drops of wax fall in to the center of the pan. Keep all flammable materials away from matches and burning candles.ġ. Determine the products of the combustion of a candle. Determine what is necessary for a candle to burn.ģ. Carefully observe and describe the characteristics of a candle flame.Ģ. In this lab, you will try to determine what is necessary for a candle for a candle to burn, and what products are formed when the candle burns.ġ. And the chemical equation says nothing about the existence of a flame.You have seen a candles burn on many occasions, but you have probably never considered the burning of a candle from a chemist's point of view. different type waxes have different chemical formulas. Note: Here the chemical formula wax can be different i.e. Since, it again turns into solid wax on cooling. 
So, it is a physical change in the burning of candles. So, we can say option D is correct.Īdditional information: On heating, candle wax melts and forms liquid wax. \įrom the above explanation we can say that formation of new products (carbon dioxide and water) takes place, by the addition of oxygen mass of product will increase and original properties of wax will change. Here, the chemical reaction of burning candle is: Also the properties of the reactants are altered. When a candle is burnt in air, the mass of the product is increased due to atmospheric oxygen which combines with the carbon and hydrogen of the candle wax during burning to produce water vapor and carbon dioxide. Burning is a chemical change that involves oxidation thus producing heat and light. The liquid wax evaporates due to the heat produced by the flame. When we light the candle, the wax present near the wick will melt. When we light the candle wax acts as fuel which is a chemical substance called carbon. Hint: Burning of candle is both physical and chemical change.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
